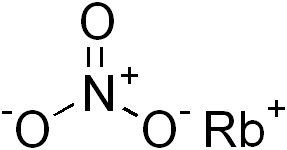Molecules with Similar Properties
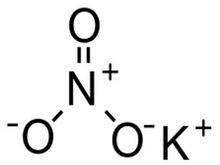
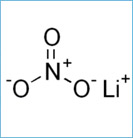
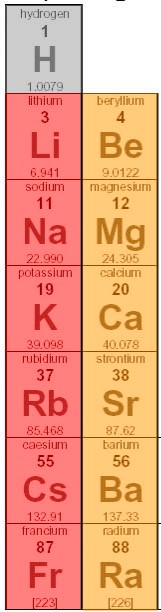
Molecules that have similar properties to Potassium Nitrate include Lithium Nitrate, Sodium Nitrate, Rubidium Nitrate, and Cesium Nitrate; these molecules are not only related by all sharing Nitrate but they are also similar because Potassium, Lithium, Sodium, Rubidium, and Cesium are all related by being in groups 1 and 2 on the periodic table. By these elements being in groups one and two, the elements both contain alkali metal in the molecules because group 1 elements are alkali metals and group 2 elements are alkali earth metals so the these molecules are also similar for that reason.
Why are the molecules similar?
A similar molecule to Potassium Nitrate is Rubidium Nitrate. Both molecules are not only alike by both bonding to Nitrate, but both Potassium and Rubidium are alkali metals, which means that each element can conduct heat and electricity well, both are softer than most other metals, and can explode if exposed to water.
What is Potassium Nitrate a part of ?
Apart from only using Potassium Nitrate to create compounds, that when ignited blow things up (Kaboom!), Potassium Nitrate is also used in so many different things you might not have expected, for example, Potassium Nitrate can be used as a food preservative in the salts that preserve the meat that people eat. It has been used this way since the middle ages, but using it this way has been almost entirely discontinued due to the inconsistent results compared to the performance of modern nitrate and other nitrite compounds. This is just one of the ways Potassium Nitrate is used today, it is also used as a fertilizer, to prepare food, and is used in some toothpastes for sensitive teeth.
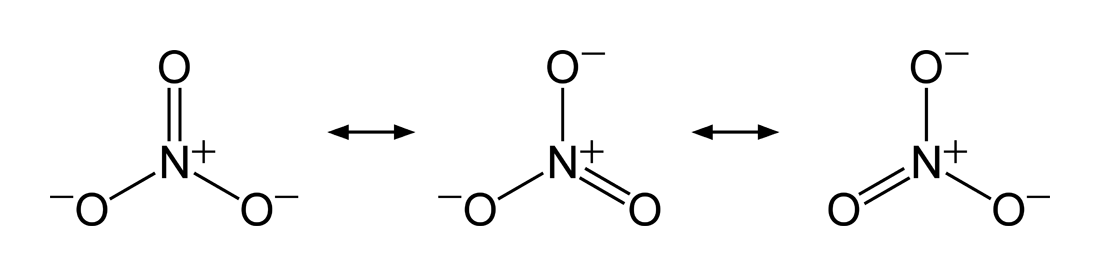
While Potassium Nitrate may not have any resonance structures that I was able to locate, Nitrate however, has three different resonance structures. The geometry for Nitrate has three different structures that are very similar. The main differences between these three structures are the way the molecule is positioned, drawn, and where each atom is placed when being constructed. Since the Nitrogen and Oxygen's are the two elements that make up the majority of the Lewis structure of Potassium Nitrate it would make sense that the resonance structures would be different mainly for the Nitrate molecule in Potassium Nitrate.
How is Potassium Nitrate used in organic compounds?
When Potassium Nitrate is mixed with organic compounds that create substances used in today’s world to produce food for people to eat, such as when fertilizer when mixed with manure, wood ashes, and common earth and organic materials such as straw, this helps to prepare the crop for growth when the rain comes. Using the organic compounds (fertilizer, manure, etc), keeps the crop moist while it is growing.
References:
http://en.wikipedia.org/wiki/Potassium_nitrate
http://www.chemicalelements.com/groups/alkali.html
http://upload.wikimedia.org/wikipedia/commons/8/81/Nitrate-ion-resonance-2D.png
http://www.westmanchemicals.com/prd/lithium-nitrate.jpg
http://upload.wikimedia.org/wikipedia/commons/4/45/Rubidium_nitrate.png
http://4.bp.blogspot.com/-ttp2OXLvsOg/UYRXmT9W1RI/AAAAAAAAACk/pY_MbgS10os/s1600/periodictable.gif
http://snohomishcoop.com/images/fertilizer.jpg
http://img1.wikia.nocookie.net/__cb20131029112301/particracy/images/3/3e/Mushroom_cloud_Forteco.jpg
http://wall4all.me/wallpaper/827707-explosions
http://www.startalkradio.net/wp-content/uploads/2012/05/hands-pouring-salt.jpg
Book Source:
Atkins, P. W. Molecules. First Printing ed. N.p.: Times, August 15, 1987. Print.
http://en.wikipedia.org/wiki/Potassium_nitrate
http://www.chemicalelements.com/groups/alkali.html
http://upload.wikimedia.org/wikipedia/commons/8/81/Nitrate-ion-resonance-2D.png
http://www.westmanchemicals.com/prd/lithium-nitrate.jpg
http://upload.wikimedia.org/wikipedia/commons/4/45/Rubidium_nitrate.png
http://4.bp.blogspot.com/-ttp2OXLvsOg/UYRXmT9W1RI/AAAAAAAAACk/pY_MbgS10os/s1600/periodictable.gif
http://snohomishcoop.com/images/fertilizer.jpg
http://img1.wikia.nocookie.net/__cb20131029112301/particracy/images/3/3e/Mushroom_cloud_Forteco.jpg
http://wall4all.me/wallpaper/827707-explosions
http://www.startalkradio.net/wp-content/uploads/2012/05/hands-pouring-salt.jpg
Book Source:
Atkins, P. W. Molecules. First Printing ed. N.p.: Times, August 15, 1987. Print.